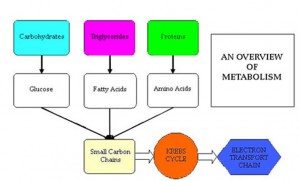
We simply think of this trial as a scientific experiment. With a little knowledge about the biochemical pathways involved in producing energy in the cell, we can make good choices about our nutrition by looking at the compounds involved in energy production pathways in order to augment them through some creative supplementation.
That's how we “hack” the mitochondria, which is the cell's powerhouse, for improved performance.
I'll cover the basics of cellular energy after the jump (fair warning, this is a lot of science. I'll try to make it as simple as possible.):
Glycolysis
 One of the basic energy pathways that our body produces energy is a process called glycolysis. This pathway (shown at right) breaks down a 6 carbon molecule known as glucose into a pair of three carbon sugars called pyruvate. Those pyruvate molecules will become very important down the chain, as you'll see in just a moment, but the take home message here is that we get energy out of this, and we end up with products that will give us even more energy a little bit later. To keep the biochemistry discussion simple, we'll refer to energy as ATP (Adenosine TriPhosphate), even though there are other chemical compounds that release energy when hydrolized (or broken apart.)
One of the basic energy pathways that our body produces energy is a process called glycolysis. This pathway (shown at right) breaks down a 6 carbon molecule known as glucose into a pair of three carbon sugars called pyruvate. Those pyruvate molecules will become very important down the chain, as you'll see in just a moment, but the take home message here is that we get energy out of this, and we end up with products that will give us even more energy a little bit later. To keep the biochemistry discussion simple, we'll refer to energy as ATP (Adenosine TriPhosphate), even though there are other chemical compounds that release energy when hydrolized (or broken apart.)
If you look closely at the diagram, two ATP are invested early on in the cycle, which turns them from ATP to ADP (Adenosine DiPhosphate) and the energy is used to change the form of the glucose molecule. This is necessary in order to cleave the molecule into two parts.
Further down the line, you'll note that 2 dinucleotide coenzymes called NAD+ (Nicotinamide Adnine Dinucleotide) are necessary to again change the form of the now 3 carbon molecules. This sets us up to extract 2 ATP from each of the 3 carbon molecules before they are turned into the pyruvate that will be fed into citric acid cycle (TCA cycle), next step of the energy production cycle.
The net yield of energy for glycolysis is 2 ATP. That's not exactly efficient.
Beta Oxidation of Fat
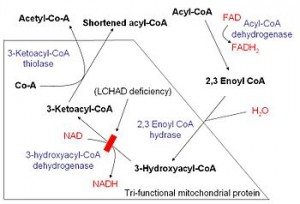 It's been written by more brilliant men than myself that the body is a fat burning machine, and they are entirely correct. We are built with a system to turn fat into huge amounts of energy. To explore that pathway, let's take the 16 carbon palmitic acid, or palmitate as an example.
It's been written by more brilliant men than myself that the body is a fat burning machine, and they are entirely correct. We are built with a system to turn fat into huge amounts of energy. To explore that pathway, let's take the 16 carbon palmitic acid, or palmitate as an example.
In the mitochondrial matrix, the goal of β-oxidation is to cleave the long carbon fatty acid into smaller parts, called acetyl CoA. Acetyl CoA is the point at which fat, carbohydrate and even some protein enters the TCA cycle. In the case of fat, we follow the following steps:
- Dehydrogenation of the C2-C3 bond, creating a double bond. (FAD cofactor)
- Hydration of C2-C3 bond, creating an L-isomer.
- Oxidation of the hydroxyl group to a keto group. (NAD+ cofactor)
- Thiolysis of the C2-C3 bond creates a 2 carbon acetyl CoA and an acyl CoA (which is the rest of the carbon chain.)
This process repeats until there are only 4 carbons left in the chain, and the final run of the cycle creates a pair of acetyl CoA molecules.
*In the case of odd numbered carbon chains the final products are an acetyl CoA and a proprionyl CoA. The proprionyl CoA is carboxylated, isomerized and then molecularly rearranged to create succinyl CoA, which enters the TCA cycle at a different point than the acetyl CoA.
The net yield of energy from 1 molecule of palmitate fatty acid is basically not realized until the acetyl CoA is fed through the rest of the process (the TCA cycle and Electron Transport Chain or ETC) but for the purposes of completeness, each β-oxidation step creates an FADH2 and NADH, which are fed into the ETC directly (bypassing the TCA cycle). There are 7 β-oxidation sites on palmitate, producing one FADH2 and one NADH at each, yielding 2 and 3 ATP each, respectively. That means 7 sites, times 5 ATP per site for 35 ATP out of β-oxidation itself. It does cost 1 ATP to start the ball rolling on β-oxidation, so the net gain is 34 ATP. That's a lot better than the 2 out of glycolysis.
But the byproducts of glycolysis and β-oxidation are what gives us a lot of energy, as you'll read below.
Protein Metabolism
Proteins can be broken down into amino acids that can also be utilized as energy. There are between 20 and 22 (depending on the text you follow) amino acids that can be introduced into the citric acid cycle as energy dense molecules. To go through where each and every amino acid enters the TCA cycle would be more tedious than it would be useful, but if you're truly curious you can read about it here. Suffice to say, since we're trying to hack our metabolism using things that are pretty readily available, I'll include a couple of them in my notes below.
The Citric Acid Cycle
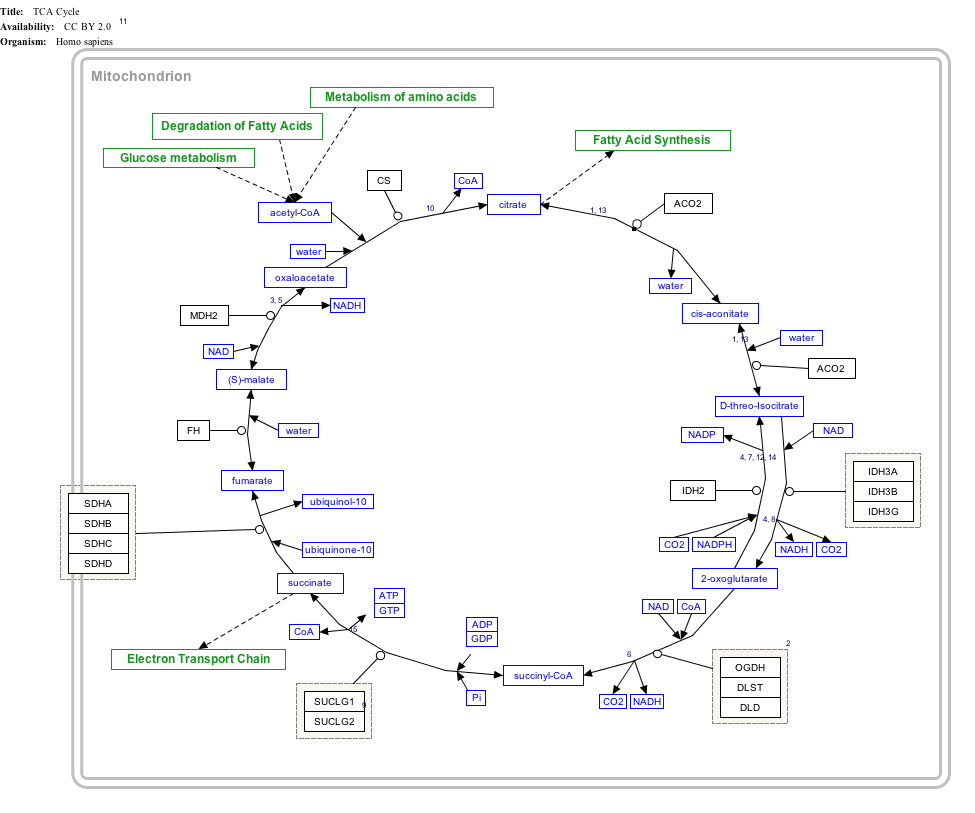
I'm just going to put this image here. Feel free to refer back to it in the following discussion:
The TCA cycle is the “big cheese” of the energy producing systems. Or really, it's more like the chubby HR lady who handles everyone's complaints. It takes the products of all different kinds of metabolism (carbohydrate, fat and protein) and it combines them into one eloquent circle of energy production. It's the HR person who makes sure everyone works together harmoniously. It also manages to spin off a number of products that will proceed into the ETC and become the massive quantities of ATP necessary to fuel working muscles.
As you can see in the upper left corner of the diagram (in the green boxes) you have the contributing pathways which provide acetyl CoA to the TCA cycle. This is generally known as where the cycle begins and is described as below:
- A 2 carbon acetyl CoA is bonded to a 4 carbon oxaloacetate via citrate synthase aldol condensation to create a citrate molecule. (requires water)
- A citrate molecule is isomerized to form cis-Aconitate (and water).
- Cis-Aconitate is isomerized to form isocitrate. (requires water)
- Isocitrate is oxidized into oxalosuccinate. (requires NAD+)
- Oxalosuccinate is decarboxylized into α-ketogluterate (a 5 carbon molecule).
- α-ketogluterate is oxidatively decarboxylized into succinyl-CoA (a 4 carbon molecule). (requires NAD+)
- Succinyl-CoA is phosphorylated into succinate.
- Succinate is oxidized into fumarate. (requires FAD and ubiquinol/CoQ10)
- Fumarate is hydrated into malate. (requires water)
- Malate is oxidized into oxaloacetate. (requires NAD+)
The whole cycle is repeated continuously to provide energy to the body and products for the ETC (which you can see breaks off between steps 7 and 8.) The net energy outcome from the TCA cycle is between 28 and 36 ATP, and that's for 1 molecule of acetyl CoA. Bear in mind that one molecule of glucose will get you 2 acetyl CoA, while one molecule of palmitate will net you 8 acetyl CoA. You can see how much more efficient fatty acid metabolism is, using simple math: so it's important to build your fat burning system properly and keep it fed (irrespective of all the other health benefits that exist with a fat fueled diet.)
Electron Transport Chain
Take a good hard look at this simple diagram. This final step in energy metabolism is a series of reactions that allow you to turn those few carbons into some epic amounts of energy.
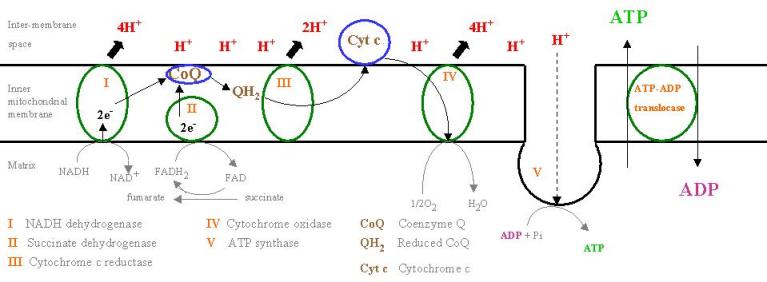
The reactants in this case are the NADH and FADH2 that was created in glycolysis, β-oxidation, protein metabolism and the TCA cycle. With each complex in the chain (the green bubbles numbered I through IV) an NADH or FADH2 is reduced, the proton (H+) is pumped out of the matrix and the electrons are shuttled through the chain. As you can see, each “turn” of the cycle dumps about 10 protons outside the matrix, and the final step results in the ATP Synthase enzyme creating an ATP molecule from an ADP and an inorganic phosphate through oxidative phosphorylation. The hydrogen moves through the ATP synthase enzyme via electrochemical gradient created by the movement of the protons.
The first set of reactions rely upon CoQ10 to mobilize the electrons between complexes I & II and complex III. Cytochrome C (a heme protein, which means it is dependent upon iron for proper formation) is responsible for the electron transfer between complex III and IV. These all rely upon on oxygen being present in the mitochondria, which relies on red blood cell iron content, oxygen binding affinity and a bunch of other factors. The biggest rate limiting factor in the ETC is simply exercise capacity: as we exceed the aerobic threshold, concentration of O2 in the cell drops. Lactate/CO2 concentration rises and we suddenly don't have the requisite oxygen to produce energy via ETC (the same goes for creating acetyl CoA from pyruvate: in the absence of oxygen, we go to a lactic acid pathway.)
As I stated earlier, the energy yield for each FADH2 is 2 ATP, the energy yield from each NADH is 3 ATP. With the ETC humming along at full capacity, you can essentially pull 5 ATP out of each “turn” of the ETC, which can run indefinitely in the presence of substrate and oxygen.
It's worth noting that it costs an ATP to transport NADH through the mitochondrial membrane, so the “net” gain of ATP from an NADH is only 2. In reality, you'll get 4 ATP for each “turn” of the ETC once you factor in the ATP used in transport.
The Bottom Line
With everything working properly, you'll gain 2 ATP out of glycolysis, 2 ATP out of the TCA cycle, and 32 ATP out of the ETC for each molecule of glucose, meaning 36 total ATP. For β-oxidation, you derive 34 ATP from the process of β-oxidation itself, 12 ATP per turn of the TCA cycle (and if you're talking 16 carbon palmitate, you have 8 turns of the TCA cycle) so 12 ATP x 8 turns = 96ATP. Add it up an that's 130 ATP from 1 molecule of palmitate.
Fat is an awesome fuel source!
The ultimate take home here is there are some commonalities between all these pathways, amongst them the coenzymes that are required to make the reactions move forward. By examining these coenzymes and substrates, we can hope to pre-load the system in order to speed up the process as we move along.
For example, a common cofactor in the TCA cycle is NAD+ which I've highlighted in green. If there's a deficiency in NAD+ generation at any point, it will stop the TCA cycle and shut off energy production, which can cause buildup of metabolites in the tissues. Supplementing the precursors of these cofactors and substrates is a simple way to boost energy and wring the last bit of performance out of your system.
Another great example of simple “problem solving” biohacking that most people will actually know very well is the CoQ10 “hack” which was discovered a number of years ago due to the rise in statin drug use. CoQ10 is an oil soluble vitamin-like substance that is required for energy metabolism (step 8 of the TCA cycle and complexes I & II of the ETC.) Synthesis of CoQ10 requires an enzyme called “HMG Co-A reductase” which just happens to be the target of the inhibitory action of statin drugs. So, the statin use decreases cholesterol (although statin use doesn't actually target the REASON for increased cholesterol, which is likely hormone imbalance or hormone signaling in the brain…more on that in the future) with a nifty side effect of inhibiting CoQ10 production. That leads to loss of energy production in the cell, muscle breakdown, pain and loss of function. The simple “hack” that gets around that is to supplement with 100-150mg of CoQ10, reducing muscle damage, pain and other unwanted side effects of statin drugs (again, without fixing the root cause, but that's still another post in itself.”
Beware though: biohacking these systems can come at a cost. Never do multiple “hacks” or supplements at a time and be sure to allow about two weeks for each trial to determine what works. Always start small and never go whole hog on a supplement. In fact, even grinding it to a powder or putting it in some oil and rubbing it on the skin is an appropriate test for sensitivity to that particular supplement.
Go slow, be careful, and be sure to check out my other articles on Biohacking for some tips on how to upgrade your fitness through some creative biochemistry.
** Please remember that the material presented above is intended for informational use ONLY. It is not intended to diagnose, treat or evaluate any condition or disease. No doctor patient relationship is implied from the presentation of this information. Consult your physician for further information about specific interventions for your medical conditions. **